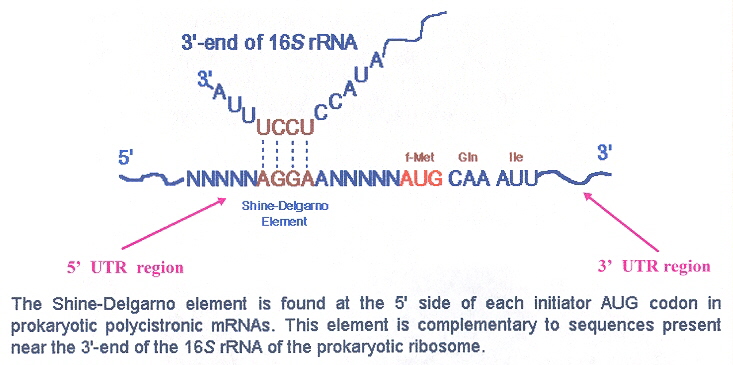
This information allows us to calculate the ribosome density on each mRNA as a proxy for the efficiency of translation initiation. Ribosome profiling is a method for deep sequencing of ribosome-protected mRNA fragments that allows us to define the position and number of ribosomes bound across the transcriptome at nucleotide resolution ( Ingolia et al., 2009). These variations across the bacterial kingdom, despite the high conservation of the ASD element on the ribosome, raise questions as to how important the SD mechanism is for ribosome recruitment. In addition, although the majority of species in the phyla Firmicutes, Actinobacteria, and Proteobacteria have high percentages of SD-containing genes, several species have low percentages, arguing that the loss of this mechanism has occurred multiple times during evolution ( Nakagawa et al., 2010 Hockenberry et al., 2017). subtilis have a high percentage of genes with SD motifs (54% and 78% respectively), there is little to no enrichment of SD motifs upstream of start codons in Bacteriodetes and Cyanobacteria ( Nakagawa et al., 2010). While well-characterized model species such as E. On the other hand, even though the ASD in 16S rRNA is almost universally conserved throughout the bacterial kingdom ( Nakagawa et al., 2010), the percentage of genes with SD motifs varies widely between species. In addition, the SD model serves as the foundation of practical bioengineering efforts ranging from optimizing expression of recombinant proteins to expansion of the genetic code ( Rackham and Chin, 2005 Salis et al., 2009). Indeed, many experiments using reporter genes showed that raising the SD-ASD affinity increases protein output, demonstrating its importance for determining translation levels ( Hui and de Boer, 1987 Jacob et al., 1987 de Smit and van Duin, 1990 Salis et al., 2009). SD motifs that differ significantly from the consensus or that are positioned too close or too far from the start codon yield lower levels of initiation. Pairing of the SD-ASD sequences can recruit 30S subunits to the start codon 5–10 nt downstream ( Steitz and Jakes, 1975). coli) referred to as the anti-Shine Dalgarno or ASD ( Shine and Dalgarno, 1974). Shine-Dalgarno motifs have the consensus sequence GGAGG and can base pair with as many as nine nt in the 3’ terminal sequence of 16S rRNA (ACCUCCUUA in E. Bound on the back of the 30S subunit, the S1 protein contains multiple RNA-binding domains that can recruit mRNA and melt secondary structures ( Qu et al., 2012), facilitating hybridization of 16S rRNA with complementary mRNA sequences colloquially known as Shine-Dalgarno motifs. Interactions of 30S subunits and single-stranded mRNA regions (especially those that are AU-rich) can be mediated through ribosome protein S1 ( Boni et al., 1991 Komarova et al., 2005). In several well-characterized examples, regions of single-stranded RNA known as standby-sites are found nearby, positioning 30S subunits in close proximity so that they can efficiently capture the start codon upon unfolding of the mRNA secondary structure ( de Smit and van Duin, 2003 Espah Borujeni et al., 2014). The kinetics of RNA folding and unfolding are also critical ( de Smit and van Duin, 2003 Espah Borujeni and Salis, 2016): some structures exist in an unfolded state for such a short period of time that 30S subunits cannot find the start codon quickly enough by diffusion alone. Thermodynamically stable secondary structures surrounding the initiation site prevent 30S recruitment ( Hall et al., 1982 de Smit and van Duin, 1990). Initiation rates vary in response to several mRNA features that determine how effectively an mRNA recruits 30S subunits to the start codon. In bacteria, a common strategy for regulating translation is to block ribosome recruitment to an mRNA through the action of small RNAs ( Altuvia et al., 1998 Majdalani et al., 1998 Storz et al., 2004), small-molecule binding riboswitches ( Winkler et al., 2002 Mandal and Breaker, 2004), and regulatory proteins ( Moine et al., 1990 Babitzke et al., 2009). Not only does initiation determine where translation occurs (and therefore which proteins are made), in most cases the rate of initiation determines the level of protein output. Bacterial ribosomes must select the correct start codons amidst a vast excess of potential sites (AUG, GUG, and to some extent UUG) that have to be ignored. 
Unlike eukaryotic ribosomes, which scan from the 5’-end of messages and generally initiate at the first start codon, bacterial ribosomes can initiate at any position along an mRNA this is a critical requirement because many bacterial mRNAs are polycistronic. Translational initiation is a critical step in the regulation of gene expression that impacts which proteins are synthesized and to what extent.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
